Clinical Evidence
Paradise® Ultrasound Renal Denervation System is designed to set a new standard in hypertension treatment.
The Paradise Ultrasound Renal Denervation (uRDN) System has been tested and proven effective across multiple clinical trials, including Recor’s RADIANCE™ global program, Real-world GPS study, the ACHIEVE and RADIOSOUND studies. With statistically significant and durable results, the Paradise System can offer a new, clinically meaningful therapy option for you and your patients.
Deliver Lasting Outcomes
3/3 randomized controlled trials (RTC)
The only RDN technology with 3/3 successful U.S. RTC, and only true resistant hypertension study found to reduce blood pressure. 1–3
Consistent and durable
The ParadiseTM Procedure showed consistent blood pressure reduction and a favorable safety profile across all pivotal studies while delivering long-term blood pressure reductions.4
Real world registry
The Global Paradise System (GPS) Registry collects real-world evidence on the performance of the Paradise Procedure.5
Triple-tested, clinically validated.
The RADIANCE global program.
Through the successful RADIANCE global program, the Paradise System became the only renal denervation (RDN) modality to meet its primary efficacy endpoint in 3 out of 3 separate powered, randomized controlled trials (RCTs) in the U.S., and the only RDN system to prove efficacy in resistant hypertension via a controlled trial.3,6
Randomized Clinical Trials
Primary endpoint at 2 months
RADIANCE-HTN SOLO
-10.8
mmHg
Office SBP reductions from baseline at 2 months1 (Off-Meds, anti-HTN medication washout) Mild to moderate hypertension (N=69)
RADIANCE-HTN TRIO
-9.0
mmHg
Office SBP reductions from baseline at 2 months3 (On-Meds, Single triple pill) Resistant hypertension. (N=136)
RADIANCE-HTN (Pooled)
-10.4
mmHg
Real-world Data
3 and 6 months among real-world EMEA GPS patients
RADIANCE-HTN SOLO
-15.7
mmHg
Office SBP reduction from baseline prospective at 3 months, uncontrolled HTN patients (OSBP ≥ 140 mmHg) treated in EMEA7 (N=137)
RADIANCE-HTN SOLO
-19.6
mmHg
Office SBP reduction from baseline prospective at 6 months, uncontrolled HTN patients (OSBP ≥ 140 mmHg) treated in EMEA7 (N=92)
Long-term Data
2-3 years
RADIANCE-HTN SOLO
-17.7
mmHg
Office SBP reduction from baseline at 3 years in (off-med study).8 (N=51)
RADIANCE-HTN TRIO
-17.3
mmHg
Office SBP reduction from baseline9,*,** (On-Meds, Single triple pill), Resistant HTN** Baseline Office SBP ≥150, mmHg at 3 years (N=30)
RADIANCE-HTN (Pooled)
-15.7
mmHg
Office SBP reduction from baseline8,** at 2 years (N=243)
* In patient population with baseline office SBP 150 mmHg.
** Baseline measurement occurred following 4-week standardization on a 3-drug single-pill.
ACHIEVE Study
8 years
24-hour Ambulatory Blood Pressure Monitoring10
-19.5
mmHg
Office Blood Pressure10
-22.1
mmHg
RADIOSOUND-HTN: Head-to-Head RCT, Optimal Results from the Main
2–3 seven-second main renal artery applications achieved comparable (numerically greater) BP reduction at 3 and 6 months versus rfRDN requiring main and distal branch ablation — with reduced fluoroscopy, contrast volume, and ablation time.11,12,**
RADIOSOUND-HTN11,12,* Primary results at 3 months.
Systolic
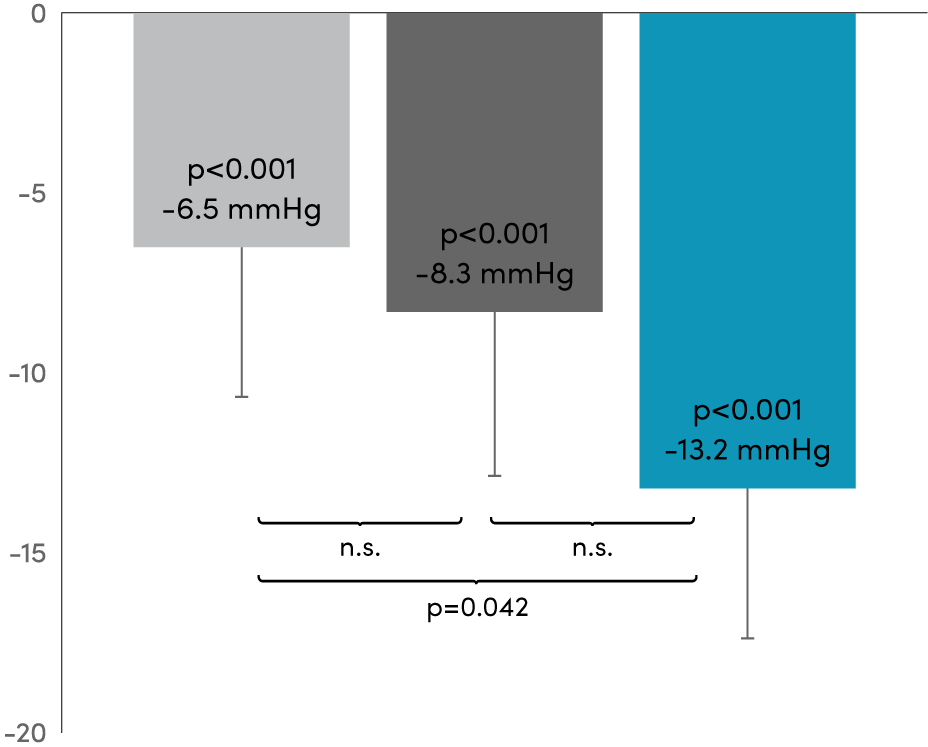
Between group difference Radiofrequency main artery vs Ultrasound -6.7 mmHg
(98.3% Cl, -13.2 to -0.2)
Diastolic
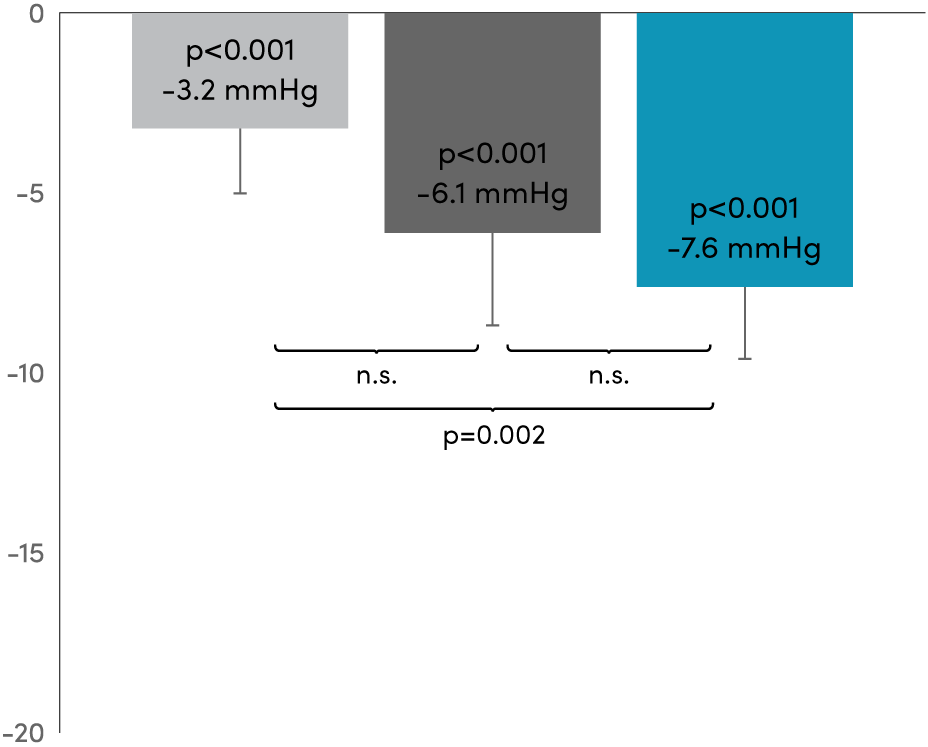
Between group difference Radiofrequency main artery vs Ultrasound -4.4 mmHg
(98.3% Cl, -8.8 to -0.52)
- Radiofrequency main artery
- Radiofrequency main artery + branches
- Ultrasound main artery
Resources
Primary endpoint publications
Reference
- Azizi M. et al., JAMA. 2023;329:651-661.
- Azizi M. et al., Lancet. 2018;391:2335-2345.
- Azizi M. et al., Lancet. 2021;397:2476-2486.
- Kirtane et al. JAMA Cardiol. 2023;8(5):464-473.
- Mahfoud F, Azizi N, Caemen J et al. 2024. Clin Res Cardiol: 113: 1375.
- V. Zeijen, ACHIEVE Study update. TCT 2023.
- Fengler, K., TCT 2025;580.
- Rader et al. EuroIntervention.2022;18:e677-e685.
- Bloch et al. Hypertens Res. 2024.
- Zeijen VJM Clin Res Cardiol. 2024.
- Fengler et al., Circulation. 2019;139:590-600.
- Fengler et al., JACC Cardivasc Interv. 2023;16:367-369.
Important Safety Information Rx Only. Brief Summary – Prior to use, please reference the Instructions for Use
Indications for Use The Paradise Ultrasound Renal Denervation System (Paradise System) is indicated to reduce blood pressure
as an adjunctive treatment in hypertension patients in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure.
Contraindications The Paradise Catheter is contraindicated in any of the following: Renal arteries diameter
- <3 mm and >8 mm
- Renal artery Fibromuscular disease (FMD)
- Stented renal artery
- Renal artery aneurysm
- Renal artery diameter stenosis >30%
- Pregnancy
- Presence of abnormal kidney (or secreting adrenal) tumors
- Iliac/femoral artery stenosis precluding insertion of the catheter
Warnings
- Failure to use the recommended balloon size may result in renal artery stenosis, dissection, perforation, aneurysm, significant vasospasm requiring intervention, ablation of unintended tissues or structures, and/or no ablation of target
tissue achieved. - Energy emission in an unintended location may result in unintended tissue damage.
- Do not move the Paradise Catheter during sonication.
- Do not sonicate in renal artery at locations with visible plaque.
- Do not deliver sonications in an overlapping arterial target zone.
Precautions
- Patients with known allergy to contrast medium may be at increased risk of hypersensitivity reactions.
- Only use specified coolant (Sterile water) for fluid supply. DO NOT USE SALINE.
- Avoid multiple balloon inflations to achieve apposition of the balloon to the renal artery wall; multiple balloon inflations may result in increased vessel trauma.
- The Paradise Catheter is for single use only. Do not resterilize or reuse. Reuse, reprocessing, or resterilization will compromise device integrity which may result in patient injury, illness, or death.
- Do not touch the Paradise Catheter balloon during sonication, as it may result in serious injury.
- The Paradise System may interfere with or adversely affect the operation of cardiac pacemakers or other active implants, unless proper precautions have been taken or managed per the manufacturer’s instructions. When in doubt regarding possible hazards, seek qualified advice and/or consult with the manufacturer(s) prior to initiating a procedure. The Paradise Catheter is a Type CF, defibrillation-proof Applied Part.
Potential risks of renal denervation procedure/response to treatment Ablation or thermal injury to vessel, adjacent tissue or other structures, Acute kidney injury, Angina, Anxiety, Arrhythmia, Atrial tachycardia, Bradycardia, Gastrointestinal complications (diarrhea, nausea, vomiting), Hypotension/ Dizziness and/or Headaches, Hypertension, Hyperhidrosis, Pain (transient abdominal, lower back), Renal failure or renal insufficiency, Renal artery aneurysm or pseudoaneurysm, Renal infarction, Renal artery dissection, or perforation, Renal artery stenosis, Vasospasm, Vasovagal response, Stroke or transient ischemic event.
Potential risks of arterial catheterization procedure Allergic reaction to contrast, Arterio-enteric fistula, Arterio-venous fistula, Bleeding, Cardiopulmonary arrest, Complications related to pain and anti-anxiety medications, Death, Deep vein thrombosis, Edema, Embolism (pulmonary, renal, peripheral vasculature, plaque), Hematuria, Infection, Myocardial infarction, Pain, Vascular access site complications (pseudoaneurysm, pain, swelling, hematoma)
Get more information about how Paradise Ultrasound RDN
when used as an adjunctive treatment for hypertension may help your hypertension patients reduce blood pressure when lifestyle modifications and antihypertensive medications are not enough.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.